Author: MSc Marcin Goras – Master of Public Health, Specialization in Emergency Medical Services
Published: 18.09.2025
Reading Time: 12-15 minutes
Introduction
Ventricular fibrillation (VF) represents one of the most critical cardiac emergencies in modern medicine, characterized by chaotic, disorganized electrical activity in the heart’s ventricles that results in immediate cessation of effective cardiac output. Emergency medicine research consistently demonstrates that VF is responsible for a significant proportion of sudden cardiac deaths worldwide, with survival rates directly correlating to the speed of recognition and intervention.
Studies indicate that ventricular fibrillation occurs in approximately 300,000-400,000 cases annually in the United States alone, making it a leading cause of out-of-hospital cardiac arrests. The condition affects individuals across all age groups, though epidemiological data suggests higher incidence rates in older adults and those with underlying cardiovascular disease. Understanding the mechanisms, recognition, and treatment of VF becomes crucial for healthcare providers, first responders, and educated members of the public who may encounter this life-threatening emergency.
Recent advances in emergency cardiovascular care have improved outcomes for VF patients, particularly through the widespread implementation of automated external defibrillators (AEDs) and enhanced cardiopulmonary resuscitation protocols. However, the narrow window for successful intervention—typically measured in minutes—underscores the critical importance of rapid recognition and immediate action.
Understanding Ventricular Fibrillation
Definition and Pathophysiology
Ventricular fibrillation is a malignant cardiac arrhythmia characterized by rapid, chaotic, and ineffective contractions of the ventricular myocardium. Electrophysiological studies define VF as irregular electrical activity with rates typically exceeding 300 beats per minute, resulting in complete loss of coordinated ventricular contraction and subsequent circulatory collapse.
The underlying mechanism involves multiple reentrant circuits within the ventricular myocardium, creating a self-perpetuating cycle of electrical chaos. Research demonstrates that these circuits form when cardiac tissue becomes heterogeneous in its electrical properties, often due to ischemia, injury, or inherited abnormalities in ion channel function.
Electrical Characteristics
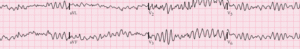
Electrocardiographic studies reveal that VF presents as irregular, chaotic waveforms without identifiable QRS complexes, P waves, or T waves. The amplitude and frequency of these waveforms can vary significantly, leading to classification systems that distinguish between coarse and fine ventricular fibrillation based on the magnitude of electrical deflections.
Coarse VF: Clinical studies suggest that coarse ventricular fibrillation, characterized by large amplitude deflections (>0.5 mV), often indicates recent onset and may be more responsive to defibrillation attempts.
Fine VF: Research indicates
that fine ventricular fibrillation, with smaller amplitude waves, may suggest prolonged arrest or depleted myocardial energy stores, often requiring additional interventions beyond simple defibrillation.
Hemodynamic Consequences
The hemodynamic impact of ventricular fibrillation is immediate and catastrophic. Studies demonstrate that effective cardiac output drops to zero within seconds of VF onset, leading to rapid loss of consciousness and, without intervention, inevitable death. Brain tissue studies indicate that irreversible neurological damage can begin within 4-6 minutes of cardiac arrest, emphasizing the critical nature of this condition.
Causes and Risk Factors
Coronary Artery Disease
Epidemiological research consistently identifies coronary artery disease as the leading cause of ventricular fibrillation, accounting for approximately 70-80% of cases in adults. The relationship between myocardial ischemia and VF involves complex interactions between reduced oxygen delivery, cellular metabolic dysfunction, and electrical instability.
Acute Myocardial Infarction: Clinical studies demonstrate that VF most commonly occurs during the acute phase of myocardial infarction, particularly in the first 24-48 hours. The mechanism involves ischemia-induced changes in cellular electrophysiology that create substrate for reentrant arrhythmias.
Chronic Ischemic Cardiomyopathy: Long-term follow-up studies indicate that patients with previous myocardial infarction and reduced ejection fraction face elevated risk for VF, particularly when left ventricular function is severely compromised.
Structural Heart Disease
| Condition Category | Specific Disorders | VF Risk Level | Key Mechanisms |
|---|---|---|---|
| Cardiomyopathies | Hypertrophic, Dilated, ARVD | High | Structural remodeling |
| Valvular Disease | Severe stenosis/regurgitation | Moderate | Pressure/volume overload |
| Congenital Defects | Complex lesions | Variable | Anatomical abnormalities |
| Inflammatory | Myocarditis, Sarcoidosis | High | Active inflammation |
Cardiomyopathies: Research demonstrates that various forms of cardiomyopathy significantly increase VF risk through different mechanisms. Hypertrophic cardiomyopathy studies show that sudden death often results from ventricular arrhythmias, particularly in young athletes.
Arrhythm
Inherited Arrhythmia Syndromes
Genetic research has identified numerous inherited conditions that predispose individuals to ventricular fibrillation:
Long QT Syndrome: Clinical studies demonstrate that patients with congenital long QT syndrome face increased risk of torsade de pointes that can degenerate into VF. Different genetic subtypes show varying patterns of triggers and risk profiles.
Brugada Syndrome: Research indicates that this sodium channel disorder creates characteristic ECG patterns and significantly elevated risk for VF, particularly during fever or exposure to certain medications.
Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT): Studies show that this condition causes exercise-induced VF in structurally normal hearts, typically manifesting in childhood or adolescence.
Metabolic and Toxic Causes
Electrolyte Imbalances: Laboratory studies consistently demonstrate strong associations between specific electrolyte disturbances and VF risk:
- Hyperkalemia: Research shows that elevated potassium levels can cause progressive cardiac conduction abnormalities culminating in VF
- Hypokalemia: Studies indicate that low potassium increases arrhythmia susceptibility through effects on cellular repolarization
- Hypomagnesemia: Clinical data suggests that magnesium deficiency predisposes to various arrhythmias, including VF
Drug Toxicity: Toxicological studies have identified numerous medications and substances that can precipitate VF:
- Proarrhythmic effects: Certain antiarrhythmic drugs paradoxically increase VF risk under specific circumstances
- Tricyclic antidepressants: Overdose studies demonstrate significant cardiotoxic effects including VF
- Cocaine and stimulants: Research shows these substances increase VF risk through multiple mechanisms including coronary spasm and direct cardiotoxicity
Environmental and Situational Factors
Hypothermia: Clinical studies indicate that severe hypothermia can trigger VF through effects on cardiac electrophysiology and cellular metabolism.
Electrocution: Emergency medicine research demonstrates that electrical injury can directly induce VF through depolarization of cardiac tissue.
Drowning and Asphyxia: Studies show that severe hypoxia can precipitate VF as a terminal rhythm in various clinical scenarios.
Clinical Presentation and Recognition
Immediate Clinical Manifestations
The clinical presentation of ventricular fibrillation is dramatic and unmistakable. Emergency medicine studies consistently describe a characteristic sequence of events that unfolds within seconds of VF onset:
Sudden Collapse: Patients experiencing VF typically collapse within 10-15 seconds of arrhythmia onset as cerebral perfusion ceases. Witnesses often describe the collapse as occurring “without warning,” though some patients may experience brief prodromal symptoms.
Loss of Consciousness: Neurological studies indicate that consciousness is lost within 8-15 seconds of VF onset due to immediate cessation of cerebral blood flow. The loss of consciousness is typically complete and immediate.
Absence of Pulse: Clinical examination reveals complete absence of palpable pulses at any site, reflecting the lack of effective cardiac contraction during VF.
Agonal Breathing: Respiratory studies show that patients may exhibit irregular, gasping respirations for several minutes after VF onset. These agonal breaths should not be mistaken for normal breathing and do not indicate adequate circulation.
Prodromal Symptoms
While VF onset is often sudden, some patients may experience warning symptoms in the hours or days preceding the event:
Chest Pain: Clinical studies suggest that some patients experience chest discomfort, particularly when VF occurs in the setting of acute myocardial infarction.
Palpitations: Research indicates that some individuals may notice irregular heartbeats or palpitations before VF onset, though these symptoms are often brief and may be overlooked.
Syncope or Near-Syncope: Studies show that patients with inherited arrhythmia syndromes may experience fainting episodes that serve as warning signs for potential VF.
Shortness of Breath: Cardiovascular research suggests that dyspnea may precede VF in patients with heart failure or acute coronary syndromes.
Physical Examination Findings
Emergency medicine training emphasizes the rapid recognition of VF through systematic assessment:
Primary Survey:
- Unresponsive patient
- Absent or abnormal breathing (agonal gasps)
- No palpable pulse at carotid or femoral arteries
- Cyanosis or pallor developing rapidly
Secondary Findings:
- Dilated pupils (developing after 2-3 minutes)
- Absence of heart sounds on auscultation
- No blood pressure obtainable
Diagnosis and Monitoring
Electrocardiographic Diagnosis
The definitive diagnosis of ventricular fibrillation relies on electrocardiographic confirmation. Emergency cardiology studies emphasize several key diagnostic features:
Rhythm Characteristics:
- Completely irregular rhythm without identifiable patterns
- Absence of discrete QRS complexes, P waves, or T waves
- Chaotic, undulating baseline with varying amplitude and frequency
- Rate typically >300 beats per minute (when measurable)
Amplitude Classification: Research has established clinical significance in differentiating between coarse and fine VF patterns, as this may influence treatment approach and prognosis.
Differential Diagnosis
Emergency medicine literature emphasizes the importance of distinguishing VF from other rhythms that may appear similar:
Ventricular Tachycardia: While VT may be rapid and wide-complex, it maintains some degree of organization and identifiable QRS morphology.
Torsade de Pointes: This polymorphic VT shows characteristic “twisting” around the baseline and typically occurs in the setting of QT prolongation.
Artifact: Technical studies indicate that loose electrodes, muscle tremor, or electrical interference can sometimes mimic VF on ECG recordings.
Asystole: Fine VF may occasionally be difficult to distinguish from asystole, requiring confirmation in multiple leads and verification of proper electrode contact.
Monitoring Considerations
Lead Placement: Research emphasizes the importance of proper electrode placement for accurate rhythm recognition during resuscitation efforts.
Artifact Prevention: Clinical studies highlight common sources of ECG artifact during CPR and strategies for minimizing interference with rhythm interpretation.
Continuous Monitoring: Emergency protocols emphasize the need for continuous ECG monitoring throughout resuscitation efforts to detect rhythm changes and guide treatment decisions.
Emergency Treatment and Management
Immediate Life Support Measures
The treatment of ventricular fibrillation follows established emergency protocols based on extensive research and clinical experience. The American Heart Association and European Resuscitation Council guidelines provide evidence-based approaches to VF management:
Recognition and Activation: Studies emphasize that rapid recognition of cardiac arrest and immediate activation of emergency medical services significantly improves outcomes. The “chain of survival” concept underscores the importance of each link in the resuscitation process.
Cardiopulmonary Resuscitation (CPR): Research consistently demonstrates that high-quality CPR provides critical coronary and cerebral perfusion during VF. Key components include:
- Compression Depth: Studies indicate optimal chest compression depth of 2-2.4 inches (5-6 cm) for adults
- Compression Rate: Research supports compression rates of 100-120 per minute for optimal effectiveness
- Minimizing Interruptions: Clinical data shows that continuous chest compressions maximize the likelihood of successful defibrillation
Defibrillation Therapy
Defibrillation represents the definitive treatment for ventricular fibrillation, with success rates directly related to time from VF onset to shock delivery:
Mechanism of Action: Electrophysiological studies demonstrate that defibrillation works by simultaneously depolarizing a critical mass of ventricular myocardium, allowing the natural pacemaker to resume control of cardiac rhythm.
Timing Considerations: Clinical studies consistently show that survival rates decrease by 7-10% for each minute delay in defibrillation, emphasizing the critical importance of rapid shock delivery.
Public Access Defibrillation: Community studies demonstrate significant improvements in VF survival rates when AEDs are readily available in public spaces and operated by trained lay responders.
Advanced Cardiac Life Support (ACLS)
Airway Management: Research supports early advanced airway placement in prolonged resuscitation efforts, though studies emphasize that airway interventions should not delay defibrillation.
Vascular Access: Clinical protocols recommend early intravenous or intraosseous access for medication administration, though drugs remain secondary to defibrillation in VF treatment.
Medication Therapy: ACLS guidelines include specific medications for refractory VF:
- Epinephrine: Studies show potential benefits through alpha-adrenergic effects that improve coronary perfusion pressure
- Amiodarone: Research indicates improved short-term survival when used for shock-refractory VF
- Lidocaine: Clinical data supports its use as an alternative antiarrhythmic when amiodarone is unavailable
Special Considerations
Hypothermic VF: Research indicates that hypothermic patients may require prolonged resuscitation efforts and multiple defibrillation attempts, as the cold heart may be resistant to electrical therapy.
Pediatric VF: Studies show that VF is less common in children but when present often indicates serious underlying pathology requiring specialized management approaches.
Pregnancy: Obstetric emergency research emphasizes the importance of perimortem cesarean delivery in pregnant patients with VF to optimize outcomes for both mother and fetus.
Post-Resuscitation Care
Immediate Post-Arrest Management
Successfully resuscitated VF patients require intensive monitoring and management to optimize neurological outcomes and prevent re-arrest:
Hemodynamic Stabilization: Critical care studies emphasize the importance of maintaining adequate blood pressure and cardiac output in the immediate post-arrest period.
Targeted Temperature Management: Research demonstrates improved neurological outcomes with controlled hypothermia (32-36°C) for 12-24 hours in comatose post-arrest patients.
Respiratory Support: Pulmonary studies indicate that mechanical ventilation with controlled oxygenation and ventilation helps prevent secondary brain injury.
Neurological Assessment and Prognostication
Glasgow Coma Scale: Neurological research provides frameworks for assessing consciousness level and tracking recovery in post-arrest patients.
Prognostic Indicators: Studies have identified various clinical, biochemical, and imaging markers that help predict neurological outcome after VF arrest.
Multimodal Assessment: Recent research emphasizes using multiple assessment tools rather than relying on single predictors for prognostication.
Cardiac Evaluation
Coronary Angiography: Cardiology studies support early cardiac catheterization in appropriate patients to identify and treat underlying coronary disease.
Echocardiographic Assessment: Imaging studies help evaluate left ventricular function and identify structural abnormalities that may have contributed to VF.
Electrophysiological Testing: Research indicates that some patients may benefit from invasive electrophysiology studies to assess arrhythmia substrate and guide long-term management.
Prevention Strategies
Primary Prevention
Risk Factor Modification: Cardiovascular prevention studies consistently demonstrate the importance of addressing modifiable risk factors:
- Hypertension Control: Research shows that optimal blood pressure management reduces VF risk through decreased cardiac remodeling
- Cholesterol Management: Lipid studies demonstrate that statin therapy reduces both coronary events and sudden cardiac death
- Diabetes Management: Endocrinological research indicates that optimal glycemic control reduces cardiovascular complications
- Smoking Cessation: Epidemiological studies show dramatic risk reduction following smoking cessation
Exercise and Lifestyle: Population studies demonstrate that regular physical activity reduces sudden cardiac death risk, though high-intensity exercise may increase acute risk in susceptible individuals.
Dietary Interventions: Nutritional research supports Mediterranean-style diets and omega-3 fatty acid supplementation for cardiovascular protection.
Secondary Prevention
Implantable Cardioverter-Defibrillators (ICDs): Clinical trials have established clear indications for ICD implantation in VF survivors and high-risk patients:
| Patient Category | Class I Indication | Evidence Level |
|---|---|---|
| VF survivors | Documented VF/VT | A |
| Ischemic cardiomyopathy | EF ≤35% on optimal therapy | A |
| Non-ischemic cardiomyopathy | EF ≤35% on optimal therapy | A |
| Genetic syndromes | High-risk features | B |
Medication Therapy: Pharmacological studies support specific medications for secondary prevention:
- Beta-blockers: Research demonstrates mortality reduction in post-MI and heart failure patients
- ACE inhibitors/ARBs: Studies show improved survival through ventricular remodeling prevention
- Aldosterone antagonists: Clinical trials indicate additional benefit in selected heart failure patients
Lifestyle Modifications: Behavioral research emphasizes comprehensive lifestyle interventions including cardiac rehabilitation programs for optimal secondary prevention.
Genetic Screening and Family Evaluation
Family History Assessment: Genetic studies recommend systematic family evaluation when VF occurs in young individuals or those without obvious structural heart disease.
Genetic Testing: Research has identified specific genetic variants that significantly increase VF risk, leading to targeted screening recommendations for at-risk family members.
Cascade Screening: Population genetics studies support systematic screening of first-degree relatives when inherited arrhythmia syndromes are identified.
Prognosis and Long-Term Outcomes
Survival Statistics
Epidemiological research provides sobering statistics regarding VF survival:
Out-of-Hospital VF: Studies consistently show overall survival rates of 8-12% for out-of-hospital cardiac arrest, with significant variation based on response time and bystander CPR rates.
In-Hospital VF: Research indicates better survival rates (15-25%) for VF occurring in monitored hospital settings where immediate intervention is available.
Witnessed vs. Unwitnessed: Clinical studies demonstrate markedly better outcomes when VF is witnessed and immediate CPR is initiated.
Factors Affecting Prognosis
Time to Defibrillation: Research consistently shows that survival decreases exponentially with time, emphasizing the critical nature of rapid intervention.
Initial Rhythm: Studies indicate that coarse VF has better outcomes than fine VF, likely reflecting shorter arrest duration and better myocardial viability.
Comorbid Conditions: Clinical research demonstrates that underlying health status significantly affects survival and functional recovery.
Age: Epidemiological studies show decreased survival rates with advancing age, though age alone should not determine treatment intensity.
Neurological Outcomes
Cognitive Function: Long-term follow-up studies indicate that VF survivors may experience various degrees of cognitive impairment, from subtle deficits to severe disability.
Quality of Life: Research shows that many VF survivors report good quality of life despite experiencing this life-threatening event.
Psychological Impact: Psychological studies indicate that VF survivors and their families often experience anxiety, depression, and post-traumatic stress symptoms requiring professional support.
Long-Term Cardiac Management
ICD Follow-up: Device studies emphasize the importance of regular ICD monitoring and appropriate programming to balance protection against inappropriate shocks.
Medication Optimization: Clinical research supports ongoing optimization of evidence-based medications for secondary prevention.
Activity Restrictions: Exercise physiology studies provide guidance on appropriate activity levels for VF survivors, balancing cardiovascular benefits against potential risks.
Special Populations
Pediatric Considerations
Ventricular fibrillation in pediatric patients requires special consideration due to different underlying causes and physiological responses:
Etiology Differences: Pediatric research indicates that VF in children is more commonly associated with congenital heart disease, inherited arrhythmia syndromes, or drug toxicity rather than coronary artery disease.
Resuscitation Modifications: Studies show that pediatric VF may require different energy dosing and medication approaches compared to adult protocols.
Long-term Outcomes: Research suggests that children who survive VF may have different long-term trajectories and require specialized follow-up care.
Pregnancy and VF
Maternal Considerations: Obstetric research emphasizes the unique challenges of managing VF during pregnancy, including modifications to standard ACLS protocols.
Fetal Considerations: Studies indicate that maternal cardiac arrest significantly compromises fetal oxygenation, making rapid maternal resuscitation critical for both patients.
Perimortem Cesarean Delivery: Research supports consideration of emergency delivery within 4-5 minutes of maternal arrest in viable pregnancies.
Elderly Patients
Comorbidity Impact: Geriatric studies show that multiple comorbidities significantly affect both the likelihood of VF survival and quality of recovery.
Treatment Decisions: Research provides frameworks for making appropriate treatment decisions in elderly patients, considering both potential benefits and burden of intensive interventions.
Family Involvement: Studies emphasize the importance of family involvement in treatment decisions for elderly VF patients.
Technology and Innovation
Advanced Monitoring Systems
Wearable Defibrillators: Recent research explores the role of wearable cardioverter-defibrillators for high-risk patients who are not candidates for implanted devices.
Remote Monitoring: Technological studies demonstrate improved outcomes with remote monitoring systems that can detect arrhythmias and alert healthcare providers.
Artificial Intelligence: Emerging research investigates machine learning algorithms for predicting VF risk and optimizing treatment protocols.
Resuscitation Technology
Mechanical CPR Devices: Clinical studies evaluate the effectiveness of automated chest compression devices in improving resuscitation outcomes.
Extracorporeal Support: Research explores the role of emergency extracorporeal membrane oxygenation (ECMO) for refractory cardiac arrest cases.
Temperature Management: Studies investigate optimal approaches to targeted temperature management using advanced cooling technologies.
Frequently Asked Questions (FAQ)
Q: What is the survival rate for ventricular fibrillation? A: Research indicates that survival rates vary significantly based on circumstances. Out-of-hospital VF has survival rates of 8-12%, while in-hospital VF shows better rates of 15-25%. The key factor is time to defibrillation—survival decreases by 7-10% for each minute of delay.
Q: Can someone survive VF without an AED or defibrillator? A: Studies show that VF rarely terminates spontaneously and virtually always requires defibrillation for survival. However, high-quality CPR can maintain some circulation and significantly improve the chances of successful defibrillation when it becomes available.
Q: How quickly does brain damage occur during VF? A: Neurological research indicates that brain cells begin to die within 4-6 minutes of cardiac arrest due to lack of oxygen. However, effective CPR can extend this window by providing some blood flow to the brain.
Q: What are the warning signs before VF occurs? A: While VF often occurs without warning, some patients may experience chest pain, palpitations, dizziness, or shortness of breath. However, these symptoms are not specific to VF and can occur with many other conditions.
Q: Can VF be prevented? A: Prevention strategies include managing cardiovascular risk factors, treating underlying heart disease, and using ICDs in high-risk patients. For those with inherited conditions, genetic counseling and family screening may be important.
Q: What happens to VF survivors long-term? A: Studies show that many VF survivors can return to normal or near-normal lives, especially with appropriate medical management including ICDs and medications. However, some may experience cognitive effects or psychological impacts requiring ongoing support.
Q: How effective are public AEDs? A: Research demonstrates that public access defibrillation programs significantly improve survival rates for out-of-hospital cardiac arrest. Studies show that bystander use of AEDs can double or triple survival rates.
Q: Can exercise trigger VF? A: While regular exercise reduces overall cardiac risk, intense exercise can trigger VF in susceptible individuals, particularly those with inherited arrhythmia syndromes or underlying heart disease. Most people can exercise safely with appropriate medical guidance.
External Resources
- American Heart Association – Sudden Cardiac Arrest
- European Society of Cardiology – Ventricular Arrhythmias Guidelines
- Sudden Cardiac Arrest Foundation
- National Heart, Lung, and Blood Institute – Sudden Cardiac Arrest
- International Liaison Committee on Resuscitation (ILCOR)
Medical Disclaimer: This article provides comprehensive educational information about ventricular fibrillation and should not replace professional medical advice, diagnosis, or treatment. The content is intended for informational purposes only and does not constitute medical recommendations or clinical guidelines. Ventricular fibrillation is a medical emergency requiring immediate professional intervention. Always seek immediate emergency medical care for suspected cardiac arrest. Treatment decisions should always be made by qualified healthcare professionals based on individual patient circumstances and current clinical guidelines. The information presented reflects current understanding of VF but medical knowledge continues to evolve, and readers should consult with healthcare providers for the most current recommendations.